|
2/12/2023 0 Comments Sp3d shape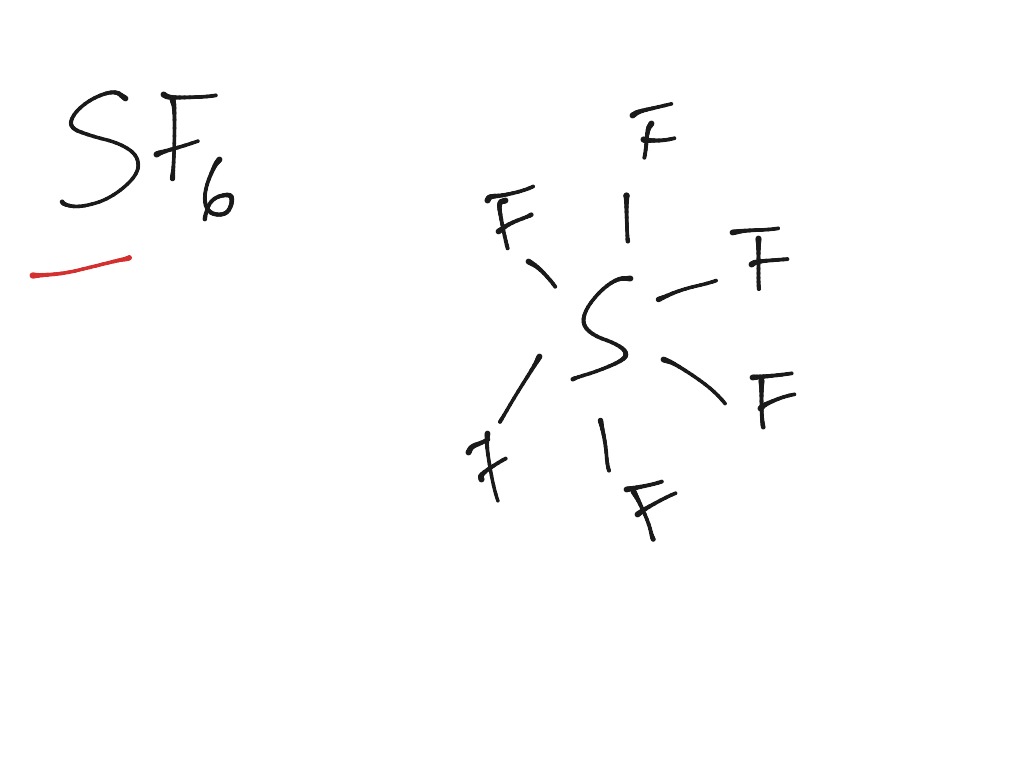
For example, CH 4 has four regions of electron density and therefore has a hybridization of sp 3 (one s, three p's = 4 letters) BF 3 has three regions of electron density and has a hybridization of sp 2 (one s, two p's = 3 letters). In sp3 the angle is 109.5 In sp3d the angles are 120 and 90. The sp 3d orbital is able to support a molecule with 5 regions of electron density, which is why BrF 3 has a hybridization of sp 3d.Īn easy way to figure out the hybridization of a molecule is that the number of regions of electron density on a molecule correspond to the number of letters in the hybridization. If a certain molecule has more than 4 regions, such as PCl 5 or XeF 4, more orbitals would have to hybridize with each other in order to create more "bonding slots." That's where the d orbitals come into play. This is because there are only 3 different p "sub-orbitals" (p x, p y, p z) and when all three hybridize with an s orbital, the highest number of hybrid orbitals that can be produced is four, meaning that the four hybrid orbitals can only support a molecule that has four regions of electron density with an unpaired electron in each hybrid orbital.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
